El Sayed H.El Ashrya. Numbers and letters correspond to the affiliation list. Click to expose these in author workspaceOpens the author workspaceOpens the author workspace. Author links open the author workspace.Laila F.Awada. Numbers and letters correspond to the affiliation list. Click to expose these in author workspace. Author links open the author workspace.Saied M.Solimana. Numbers and letters correspond to the affiliation list. Click to expose these in author workspaceb. Numbers and letters correspond to the affiliation list. Click to expose these in author workspace. Author links open the author workspace.Mohamed N.Abd Al Moatya. Numbers and letters correspond to the affiliation list. Click to expose these in author workspace. Author links open the author workspace.Hazem A.Ghabbourc. Numbers and letters correspond to the affiliation list. Click to expose these in author workspace. Author links open the author workspace.AssemBarakat
Abstract
Reaction of 4-amino-3-methyl-1,2,4-triazole-5-thione with some substituted benzaldehydes in presence of catalytic amount of hydrochloric acid afforded the respective Schiff's bases 3a-e. Computational studies using DFT incorporating the B3LYP/6-311G(d,p) level of theory is used to predict the stability of the possible tautomers. Molecular modeling, natural charge calculations, NMR, Frontier molecular orbitals and electronic spectra were investigated. Analysis of the thermodynamic parameters of the thione-thiol tautomeric reactions of these derivatives was used to predict the tautomers stability. The thione tautomer is the most favored form in gas phase and in solution whatever the nature of solvent used. The electronic spectra were assigned based on the TD-DFT calculations. The GIAO NMR chemical shifts correlated well
with the experimental data.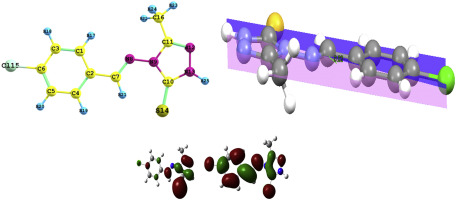

 English
English